Your trusted CDMO partner based in Europe
Specialized in small-molecule dosage-form development, reformulation and EU-GMP manufacturing.
Specialized in small-molecule dosage-form development, reformulation and EU-GMP manufacturing.

A reliable partner for global markets

Human and Veterinary

The agility of a mid-sized CDMO

A reliable partner for global markets

Human and Veterinary

The agility of a mid-sized CDMO
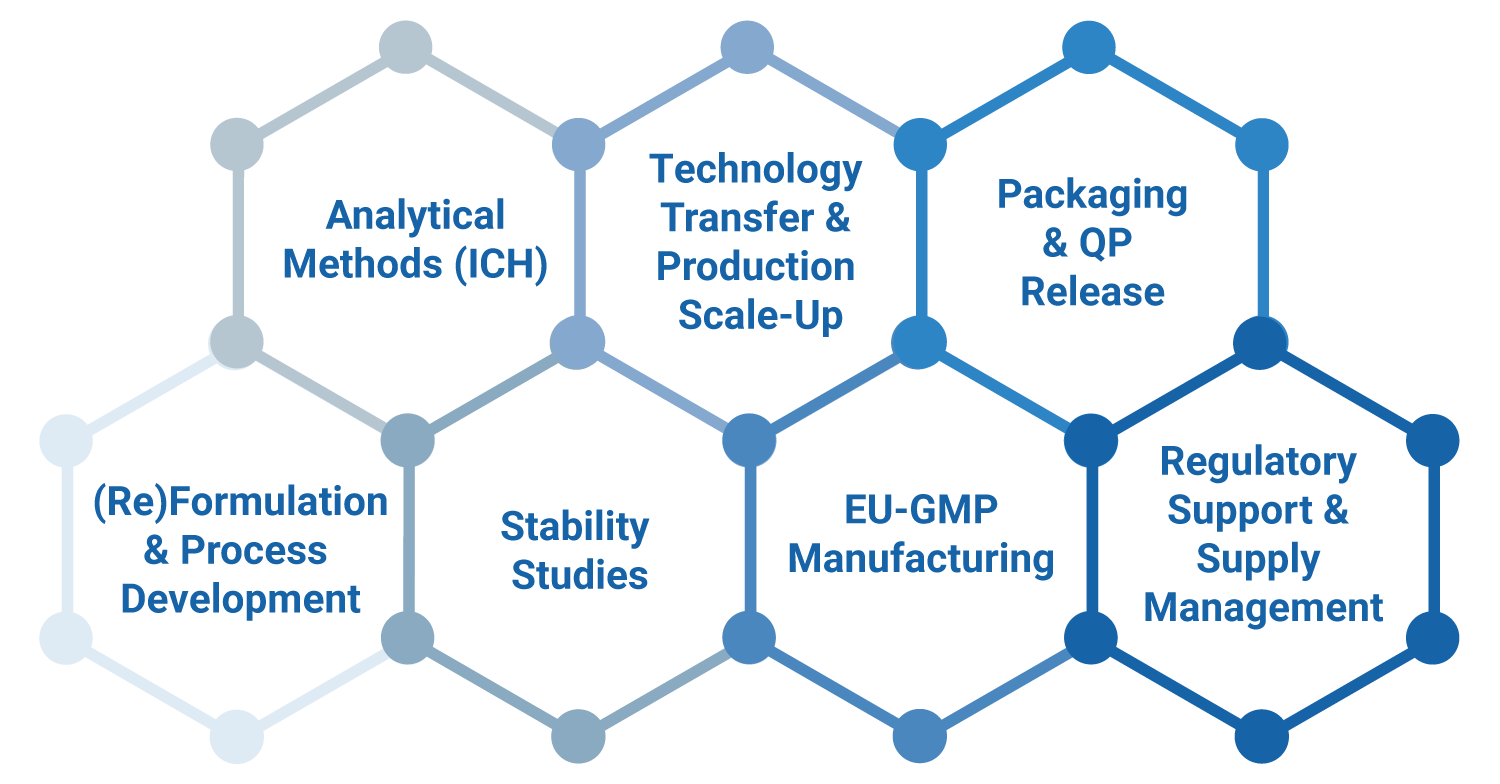
Whether you need support at the beginning, mid-way or close to commercial production, we slot into your process and keep your project moving smoothly.
70% of our clients are based in Europe and 30% overseas. We know what it takes to run international projects efficiently. Ready to explore how we can support your next phase? Talk to our Experts!
We provide end-to-end support in formulation, reformulation, analytical services, scale-up and EU-GMP manufacturing, for both sterile and non-sterile products in human and animal health.
We stand out for our know-how in niche dosage forms, both sterile and non- sterile, such as powders for injection, nasal sprays and ophthalmic ointments.
BePharBel Manufacturing has strong expertise in sterile drug manufacturing. Our aseptic area is a controlled atmosphere zone (A class) in compliance with the EN/ISO14644-1 standard.



Tablets
Capsules
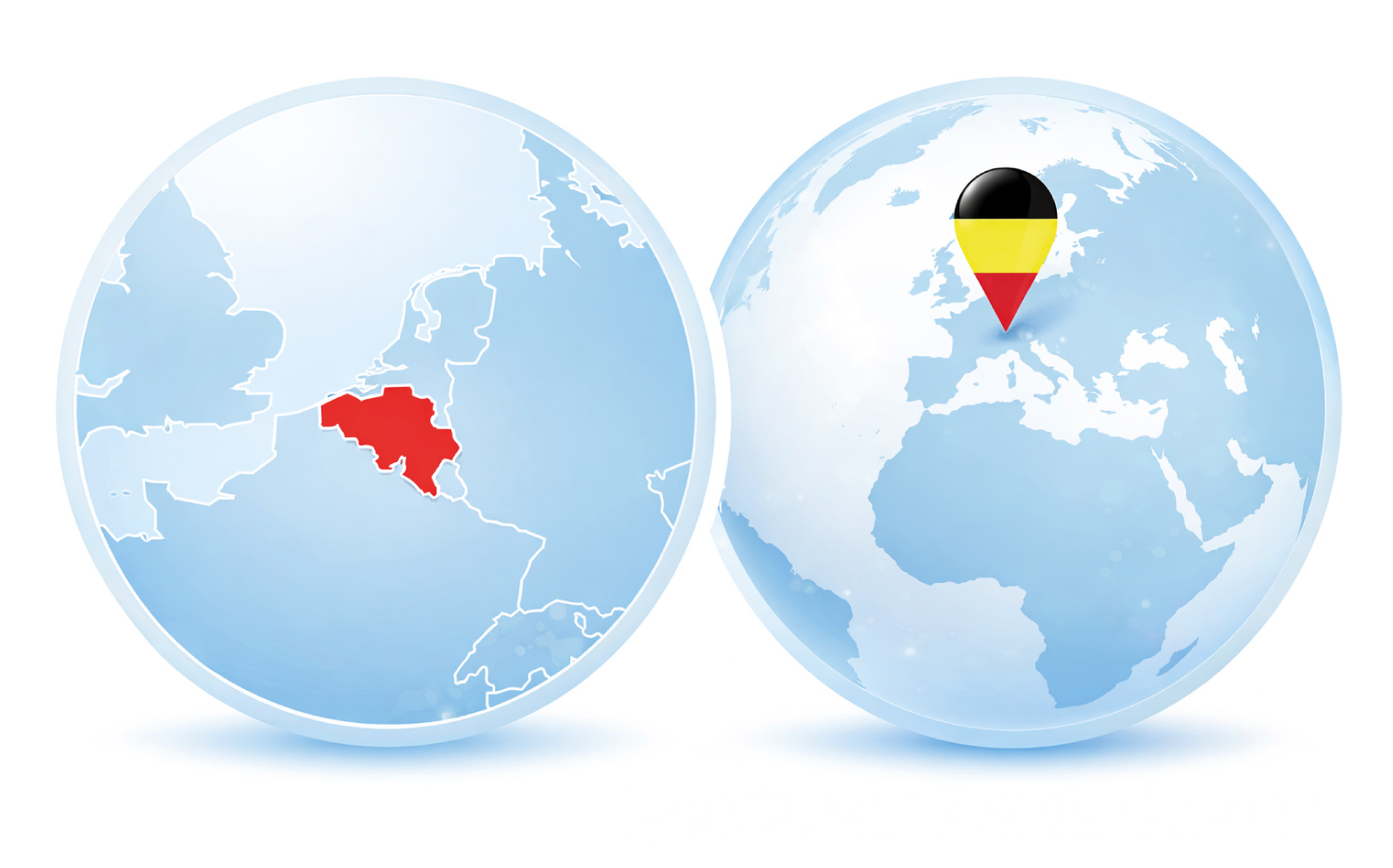
By choosing BePharBel Manufacturing as your CDMO partner, you enter a leading European pharmaceutical ecosystem.
Belgium is among Europe’s top pharmaceutical producers and exporters, offering a dense network of pharma companies, research institutions and logistics platforms.
BePharBel Manufacturing benefits from MRAs with Canada, Japan, and other markets, enabling you to supply multiple regions from a single site.
Within this ecosystem, we collaborate with academic and industry partners that reinforce our R&D and support our visibility in Europe.



Our 12,000 m² EU-GMP site in Courcelles brings together R&D, formulation, analytical methods, pilot scale and full commercial production on one EU-GMP site.
Your project stays with the same equipment, documentation and QA/QP team from lab scale to commercial batches, avoiding inter-site transfers, duplicate validation work and additional audits.
12.000m2
BePharBel Manufacturing is an independent Belgian SME with a mix of public and private shareholders and no venture-capital ownership. This stable structure supports a long-term vision aligned with the long lifecycles of pharmaceutical development. We prioritise partnership over one-off projects and rely on transparent communication to build durable relationships.
Our multidisciplinary team brings solid experience in pharmaceutical development, manufacturing, quality and regulatory affairs. We work closely with you to understand your product, anticipate challenges and find pragmatic solutions.


